Consent forms are your first step in building trust, transparency, and accountability. Most people ignore consent forms until there’s a problem. Maybe someone didn’t understand what they agreed to, or the form missed key details. Today, with data and decisions under the spotlight, using the right consent form is a must. It’s essential to keep ourselves away from any legal, ethical, or communication mishaps by laying everything out clearly from the start.
This article helps to get to know more on different types of consent forms, best practices, and how to create one using a free form builder.
Understanding consent forms
A consent form is a document that confirms someone agrees to something knowingly, voluntarily, and with complete understanding. It could be about sharing data, participating in a study, receiving treatment, or even being featured in a video. It protects both parties by laying out expectations and responsibilities upfront.
Verbal vs written vs digital consent
Consent can be given in different ways:
- Verbal works for quick or informal agreements, but is hard to prove.
- Written (paper-based) is reliable but not always convenient.
- Digital forms created using a form creator are easy to store, send, and sign, especially for remote or large-scale use.
When should you use a consent form?
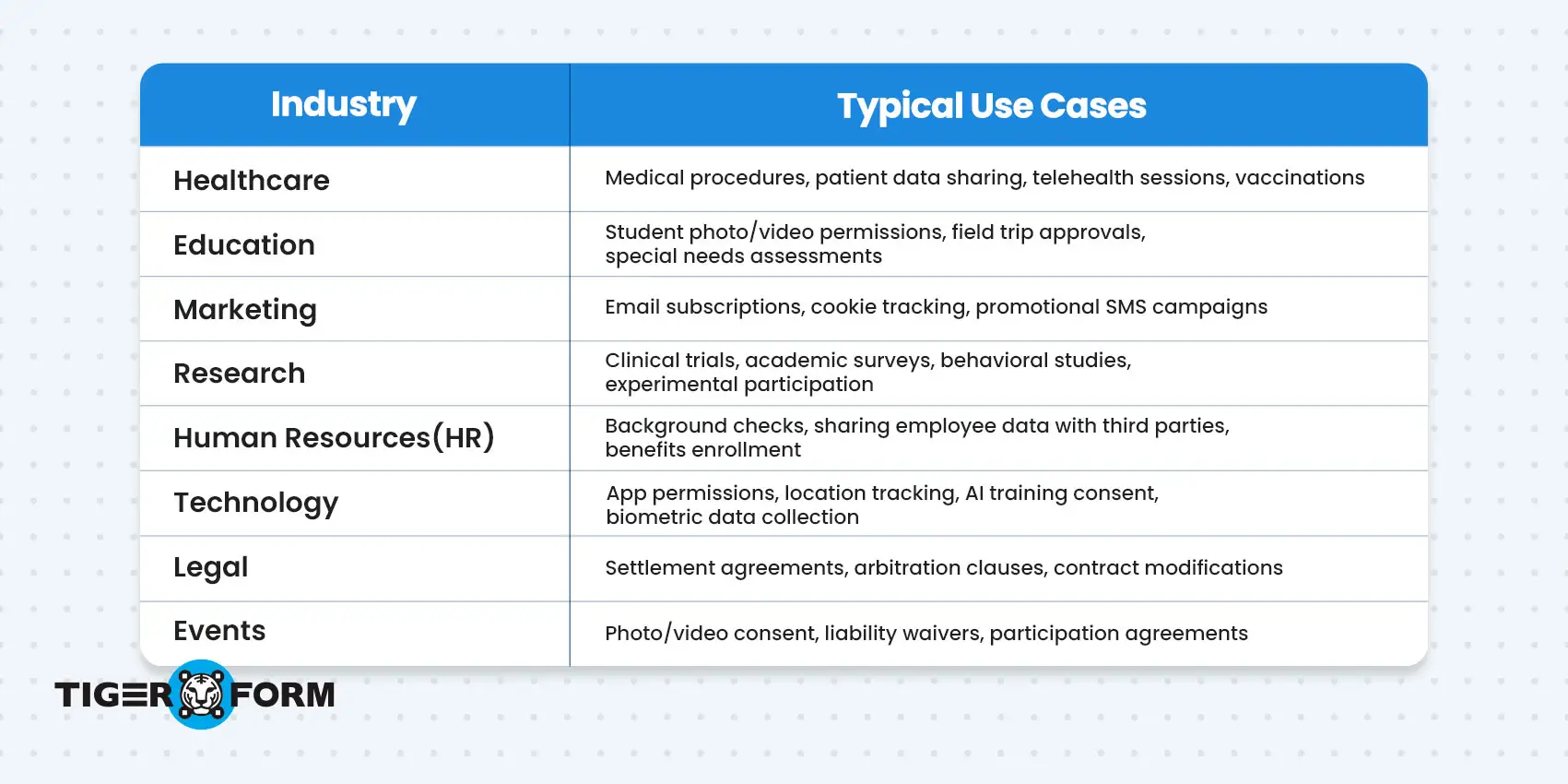
Use a consent form anytime you collect personal information, request participation, or ask someone to make a decision that affects their rights, data, or well-being. Whether it’s through a research study, job application, email subscription, or any other data collection method, if there’s something to agree to, you need consent.
Below is a table showing when and where consent forms are typically used across different industries:
Common types of consent forms
1. Informed consent form
An Informed consent form is essential in clinical trials, healthcare procedures, and academic research. It ensures participants understand exactly what they’re agreeing to, what will happen, the risks involved, potential benefits, and their right to withdraw at any time. Without it, you’re not just legally exposed, you’re breaching ethical trust.
Perfect for: Doctors, hospitals, clinical researchers, academic institutions, psychologists
Key fields to include:
- Participant’s name and contact information
- Description of the procedure or study
- Purpose and objectives
- Potential risks or side effects
- Expected benefits
- Alternatives (if any)
- Right to refuse or withdraw
- Confidentiality clause
- Contact info for questions
- Signature and date
2. Implied consent form
An Implied consent form is typically used when individuals consent through their actions rather than a physical signature. It’s common in low-risk, anonymous activities like surveys, online polls, user research, or public observations, especially when identifying information isn’t collected. The form makes it clear that by proceeding, the participant understands what’s involved and agrees to take part.
Perfect for: Academic researchers, UX researchers, educational institutions, nonprofit studies, marketing teams running anonymous surveys
Key fields to include:
- Study or activity title
- Description of what participation involves
- Purpose and intent
- Any minimal risks
- Confidentiality assurance
- Voluntary nature of participation
- Contact info for questions
- Implied consent acknowledgment
- Date
3. Explicit consent form (Express or active consent)
An Explicit consent form is used when the participant or user must actively agree to a very specific action, such as receiving marketing emails, sharing sensitive information, or allowing third-party data transfers. It’s not assumed or implied; consent must be clearly given, usually by checking a box or signing a form.
This type of consent is legally required under privacy laws like GDPR or HIPAA when processing personal or sensitive data, making it one of the most important forms in digital services, healthcare, and marketing.
Perfect for:
Email marketing, app permissions, data sharing agreements, HIPAA-covered healthcare services, biometric data collection, clinical trials, financial platforms
Key fields to include
- Full name and contact details
- Description of what’s being agreed to
- Purpose of the request
- Risks or consequences of agreeing/not agreeing (if applicable)
- Statement that consent is voluntary
- Consent checkbox
- Signature and date
- Link to privacy policy or terms (optional but recommended)
4. Parental/guardian consent form
A Parental or guardian consent form is used when minors are involved in any activity, whether it’s a school trip, medical procedure, online research, or extracurricular program. Since children can’t legally provide informed consent themselves, this form ensures their parent or legal guardian understands what’s happening and approves the child’s participation.
Perfect for: Schools, camps, hospitals, pediatric clinics, researchers working with minors, and educational app developers
Key fields to include:
- Child’s name and age/date of birth
- Parent or guardian’s name and contact information
- Description and purpose of the activity or study
- Potential risks and expected benefits
- Emergency contact details
- Medical conditions or special accommodations (if applicable)
- Explicit statement of parental/guardian consent
- Signature and date
5. Medical consent form
A Medical consent form gives healthcare providers legal and ethical permission to perform specific medical procedures or treatments. It confirms that the patient (or their guardian) understands the diagnosis, treatment options, risks, and alternatives and agrees to move forward. This form is crucial for surgeries, diagnostic tests, vaccinations, emergency care, and any treatment involving patient rights or bodily autonomy.
Perfect for: Hospitals, clinics, diagnostic labs, dentists, physiotherapists, telemedicine providers, caregivers of minors or elderly patients
Key fields to include:
- Patient’s full name and contact information
- Description of the procedure or treatment
- Reason for the treatment
- Risks and possible side effects
- Potential benefits
- Alternative treatments or refusal consequences
- Right to ask questions and withdraw
- Emergency contact
- Signature of patient or guardian
- Signature of medical provider
- Date of consent
6. Research consent form
A Research consent form is used when someone agrees to participate in academic or scientific studies like observational, experimental, or survey-based. It ensures participants know what the study involves, how data will be collected, how long it will take, any risks or discomforts, and how their privacy will be protected. It also outlines their right to skip questions or leave the study at any time.
Perfect for: Universities, independent researchers, think tanks, NGOs, clinical trial coordinators, social science projects
Key fields to include:
- Participant’s full name and contact details
- Study title and purpose
- Duration of the study
- Description of participation (tasks, time commitment)
- Risks or discomforts
- Benefits (if any)
- Voluntary nature of participation
- Data handling and privacy policy
- Contact for questions or complaints
- Signature of participant and researcher
- Date
7. Data/privacy consent form
A Data or privacy form is used when you collect, store, or share a person’s personal data, whether for marketing, analytics, HR, user registration, or any digital service. This form outlines exactly what data is being collected, how it will be used, and the user’s rights under privacy laws like GDPR, HIPAA, or local regulations. It helps you stay transparent, avoid legal trouble, and build trust.
Perfect for:
SaaS companies, eCommerce platforms, HR departments, mobile apps, survey tools, healthcare providers, and financial institutions
Key fields to include
- Full name and contact info
- Description of what personal data is collected
- Purpose of data collection
- How data will be used/shared
- Legal basis (consent, contract, compliance, etc.)
- Data retention period
- Rights of the individual (e.g. to access, correct, delete data)
- Consent checkbox
- Signature and date

How to create a consent form using a free form creator
Step 1: Choose the right online form creator
Before anything else, pick a online form creator that’s reliable, easy to use, and built with privacy in mind. TIGER FORM is a great choice here as it is beginner-friendly and has QR code integration. The platform also offers a wide range of templates to choose from.
Step 2: Set your form title and intro
Start by naming the form clearly something like Patient Consent Form. Use the intro field to explain what the form is about, who it’s for, and how the responses will be used.
Step 3: Add consent fields
Add relevant fields according to your form. In your form online builder, you can even group sections into collapsible blocks so users don’t feel overwhelmed.
Step 4: Design and customize
Customize fonts, colors, and layout to match your brand. Consent form templates give you complete control to speed things up.
Step 5: Preview and test
TIGER FORM’s live preview shows you exactly what users will see on desktop and mobile. Run a quick test to ensure everything works especially required fields and signature capture.
Step 6: Share and distribute
Once your form is ready, publish it with one click:
– Share via secure link
– Generate a QR code instantly for print or in-person use
– Embed it directly into your website or landing page
Step 7: Manage responses securely
All submissions are stored securely in your TIGER FORM dashboard. You can:
– Export to Excel or sheets
– Get notified by email
– Set access permissions for your team
– Ensure compliance with privacy regulations (GDPR, HIPAA, depending on use)
You can unlock more features while creating a form by exploring TIGER FORM guide.
Best practices for creating a consent form
A solid consent form does more than meet legal requirements. It builds trust, transparency, and accountability.
Here’s how to make sure your form is clear, ethical, and effective.
1. Be clear and direct
If someone can’t understand your form in one read, it’s too complicated. Use short sentences, active voice, and simple terms. Confusing language can lead to misunderstandings or invalid consent.
How to do it well:
- Replace legalese with everyday language.
- Use headings and bullet points to organize information.
- Avoid passive constructions like “you may be subjected to.”
- Write as if you’re talking directly to the participant or user.
2. Tell people exactly what they’re agreeing to
Full disclosure builds informed consent and prevents legal/ethical backlash later.
Spell out:
- What’s being asked of them
- Why it’s being done
- What will happen to their data or responses
- What the risks and benefits are
3. Highlight the right to refuse or withdraw
People must know they can say “no” at any time before or during participation without facing any negative consequences. This is a legal and ethical non-negotiable in most sectors, including research, healthcare, and education.
4. Make mandatory fields obvious
Don’t frustrate people by making them guess what’s required. Make your form accessible and user-friendly. Worse, if they miss key sections, the consent might not hold up legally.
How to do it well:
- Use asterisks (*) for required fields.
- Keep the form short and focused every field should have a clear purpose.
- Use conditional logic (showing certain questions only when relevant) to avoid clutter.
5. Keep a copy for records
Participants should be able to refer back to what they agreed to. It protects both sides. Having a record prevents disputes, especially in long-term studies or agreements.
6. Match format to context
Not all forms need the same level of depth. A marketing opt-in form and a surgical consent form don’t require the same structure but both need clarity. Overloading low-stakes forms can reduce conversion. Under-explaining high-risk ones can lead to non-compliance.
How to do it well:
- Use toggle sections or collapsible info blocks for longer forms.
- For web forms, keep the most important parts visible above the fold.
7. Use accessible design
Good consent isn’t just about the words; it’s about who can understand and interact with the form. You might be collecting consent from people with low literacy, disabilities, or different language backgrounds.
How to do it well:
- Use readable fonts (no smaller than 12pt) and high contrast.
- Use screen reader–compatible form elements.
- Offer translated versions or visual aids where necessary.
Build trust with every click — Create your consent form now
A good consent form shows people exactly what they’re signing up for—and why they should trust you. A clear, well-structured consent process builds transparency. It shows participants or clients that you respect their rights, their data, and their ability to make informed choices.
Take a moment to audit your existing consent forms. Are they clear? Up to date? Do they match the way you actually collect and store information? If not, it might be time to refresh your workflow and a tool like TIGER FORM can help you do that quickly and securely.
Visit our website to start with a template today.
FAQs
1. What is a sample consent form?
A sample consent form is a ready-made template that shows how to ask for permission clearly. It usually includes sections like the purpose, risks, benefits, rights, and a place for the participant’s signature. These samples help you build compliant, easy-to-understand forms without starting from scratch.
2. What are the 4 areas of consent?
The four key areas of consent help define when and how consent should be obtained, especially in legal, medical, and ethical contexts:
Specific – Consent is given for a particular action or purpose, not a blanket agreement for anything and everything.
Voluntary – The person gives consent freely, without pressure, manipulation, or coercion.
Informed – They fully understand what they’re agreeing to—including the purpose, risks, benefits, and alternatives.
Capacity – They’re mentally and legally capable of giving consent (e.g., of sound mind, of legal age).
3. What are the 5 C’s of Consent?
The 5 C’s of Consent stand for Choice, Consent, Clarity, Consistency, and Control. Together, they ensure that consent is freely given, clearly understood, properly recorded, and can be changed or withdrawn at any time.